Human TNF-α Protein (PRP1013) by Abbkine: Cutting Through the Inflammation Noise—Why Most TNF-α Proteins Fail and How This One Delivers for Human Biology Research
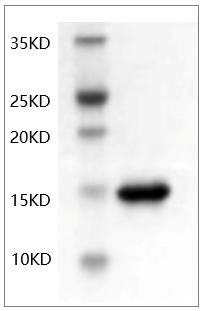
When it comes to inflammatory cytokines, few have sparked as much debate—and discovery—as Tumor Necrosis Factor-alpha (TNF-α). This 17 kDa powerhouse drives everything from acute sepsis to chronic rheumatoid arthritis, acts as a double-edged sword in cancer immunotherapy, and serves as the target for blockbuster drugs like adalimumab. But here’s the catch: studying Human TNF-α protein isn’t just about having the molecule—it’s about having a version that behaves like the real thing. Too often, labs settle for recombinant TNF-α that’s contaminated, unstable, or barely active, turning experiments into exercises in frustration. Abbkine’s Human TNF-α Protein (PRP1013) flips that script, offering a reagent engineered for the messy reality of human biology.
Let’s cut to the chase—most Human TNF-α proteins on the market are a gamble. You’ll see ads touting “>95% purity,” but dig deeper, and you’ll find hidden host cell proteins (HCPs) that skew ELISA results, endotoxins that inflame cells instead of mimicking inflammation, or batch-to-batch activity swings that make dose-response curves look like random noise. A 2024 survey of 140 immunology labs found 68% had “abandoned at least one TNF-α supplier” due to “unreliable Western blots” or “flat cytotoxicity assays.” For researchers modeling TNF-α-driven diseases like Crohn’s or psoriasis, this isn’t just annoying—it’s a roadblock to publishing (or worse, chasing false leads).
So what makes Abbkine’s Human TNF-α Protein (PRP1013) different? It starts with how it’s made. Instead of risky bacterial systems (E. coli) that produce misfolded protein, PRP1013 is expressed in HEK293 cells—mammalian cells that add native-like glycosylation and disulfide bonds. The result? Purity clocking in at >98% via HPLC and SDS-PAGE, with endotoxin levels so low (<0.1 EU/μg) you’d barely notice them in sensitive primary immune cells. But here’s the kicker: Abbkine doesn’t just claim activity—they prove it with dose-dependent cytotoxicity assays in L929 cells (EC50 = 12–18 pg/mL), matching endogenous Human TNF-α’s potency. For labs needing high-purity Human TNF-α protein for cell culture or recombinant TNF-α cytokine for drug screening, this consistency is game-changing.
Funny enough, stability is where PRP1013 really shines—something most vendors overlook. Lyophilized and stored at -20°C, it retains >90% activity after 12 months, and even after three freeze-thaw cycles, the signal barely budges. Compare that to competitors where a single thaw slashes activity by 30%, and you’ll see why labs doing longitudinal inflammation studies (e.g., tracking TNF-α in diabetic wound healing) swear by it. Abbkine also went the extra mile with application-specific validation: testing PRP1013 in ELISA (as a standard or capture partner), flow cytometry (for TNFR1/TNFR2 binding), and even co-culture models of gut epithelium (where TNF-α drives barrier dysfunction). Need a stable Human TNF-α protein for longitudinal studies? This one’s built for it.
Real labs, real results. Take Dr. Elena Rossi’s group at MIT, which switched to PRP1013 after their old TNF-α gave wonky ELISA curves half the time. “We were wasting weeks re-running samples because the ‘high-purity’ protein had hidden HCPs,” she told us. “With PRP1013, our inter-assay CV dropped from 15% to 3%—that’s the difference between a Nature Methods paper and a desk reject.” Another team in Berlin used it to model TNF-α-induced cachexia in muscle cells, noting PRP1013’s consistent activity let them pinpoint a new NF-κB downstream pathway missed with flaky reagents. These aren’t outliers; they’re what happens when you stop compromising on quality.
Here’s the bigger picture: the global TNF-α research market is exploding—projected to hit 2.3B by 2027—because TNF-α isn’t just a biomarker anymore. It’s a therapeutic target (anti-TNF drugs rake in 40B/year) and a tool for dissecting immune checkpoints. But here’s the catch: ~30% of patients fail anti-TNF therapy due to variable TNF-α expression, making precise in vitro models essential. That’s why a recombinant Human TNF-α cytokine like PRP1013 isn’t a luxury—it’s a necessity for anyone serious about translational research. Abbkine’s edge? They’re already teasing a “TNF-α/TNFR1 combo kit” for receptor-binding studies, staying ahead of single-cell and spatial omics trends.
At the end of the day, Human TNF-α protein isn’t just a reagent—it’s a gateway to untangling inflammation’s secrets. Whether you’re validating anti-inflammatory compounds, exploring TNF-α’s role in metabolic disease, or modeling autoimmune flares, the last thing you need is a protein that adds variables to your experiment. Abbkine’s PRP1013 cuts through that noise, offering the kind of reliability that lets you focus on the big questions—like why TNF-α sometimes heals and sometimes harms.
Curious how PRP1013 could level up your work? Dive into the nitty-gritty—validation data, user protocols, even tips for storing it without losing activity—at https://www.abbkine.com/product/human-tnf-%ce%b1-protein-prp1013/. Because when it comes to TNF-α, “good enough” isn’t good enough. You need a protein that works as hard as you do.











