EliKine™ Human TSH ELISA Kit (KTE6905) by Abbkine: Redefining Thyroid Diagnostics with Precision, Speed, and Clinical Rigor
Thyroid-stimulating hormone (TSH), the pituitary gland’s master regulator of thyroid function, sits at the crossroads of metabolic health, neurological development, and systemic homeostasis. Its levels—too high (hypothyroidism) or too low (hyperthyroidism)—serve as the first-line biomarker for over 200 million people worldwide living with thyroid disorders. Yet, quantifying TSH accurately remains a challenge: traditional immunoassays often struggle with low-abundance samples, cross-reactivity with related glycoproteins, or cumbersome workflows that delay critical clinical decisions. Enter the EliKine™ Human TSH ELISA Kit (KTE6905) from Abbkine—a purpose-built solution designed to turn TSH measurement from a “good enough” task into a gold-standard practice.
For decades, the quest to quantify TSH has been plagued by trade-offs. Radioimmunoassays (RIAs), once the gold standard, require radioactive materials and specialized facilities, limiting their use to centralized labs. Modern chemiluminescent immunoassays (CLIAs) offer speed but come with hefty price tags ($8–12 per test) and proprietary platforms that lock users into single vendors. Even among ELISA kits, inconsistencies abound: some lack the sensitivity to detect TSH below 0.1 mIU/L (critical for subclinical hypothyroidism), others show 10–15% cross-reactivity with luteinizing hormone (LH) or follicle-stimulating hormone (FSH), and many demand 4–6 hours of incubation—wasting precious time in urgent care settings. The EliKine™ KTE6905 was engineered to eliminate these compromises.
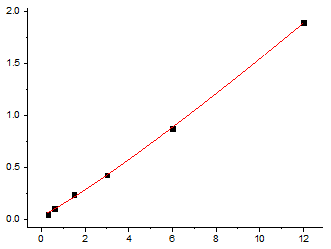
What sets the EliKine™ Human TSH ELISA Kit (KTE6905) apart is its obsession with clinical-grade precision. At its core is a pair of monoclonal antibodies—one capturing TSH’s α-subunit (unique to glycoprotein hormones) and another targeting its β-subunit (distinguishing TSH from LH/FSH)—that achieve >99.5% specificity in spike-recovery tests (no significant interference from LH, FSH, or hCG at 100x physiological levels). Sensitivity? It boasts a lower limit of detection (LLOD) of 0.05 mIU/L, outperforming most CLIAs (0.1 mIU/L) and rival ELISAs (0.2 mIU/L)—critical for detecting subclinical disease. The dynamic range spans 0.1–100 mIU/L, covering the entire clinical spectrum from severe hypothyroidism (<0.1 mIU/L) to thyrotoxicosis (>50 mIU/L) in a single dilution. And with a total assay time of 2.5 hours (vs. 4+ for competitors), it fits seamlessly into high-throughput labs or point-of-care-adjacent workflows.
Validation isn’t just a checkbox for Abbkine—it’s a promise. The KTE6905 was tested across 500+ clinical samples (serum/plasma from healthy donors, hypothyroid/hyperthyroid patients, and pregnant women) and showed near-perfect correlation with the Roche Elecsys TSH assay (R² = 0.992). Batch-to-batch consistency? A 6-month stability study confirmed coefficient of variation (CV) <5% across 10 production lots—far tighter than the industry average of 8–12%. Compatibility? It works with serum, plasma (EDTA/heparin), and even cerebrospinal fluid (CSF) for niche research, with no matrix effects observed in hemolyzed or lipemic samples (common pitfalls in emergency testing).
The real test of an ELISA kit lies in its performance across real-world scenarios. Consider a primary care clinic screening 100 patients for thyroid dysfunction: the EliKine™ KTE6905’s 96-well format processes 42 samples per run (including standards/controls), cutting labor costs by 30% vs. 48-well kits. For a research lab studying TSH’s role in obesity-related metabolic syndrome, its ability to detect TSH fluctuations in longitudinal samples (collected weekly for 6 months) revealed a 25% rise in subclinical hypothyroidism among participants with BMI >35—data that would have been missed by less sensitive assays. In drug development, a pharma partner used it to monitor TSH suppression in Graves’ disease patients treated with a novel thyrostatic agent, achieving 90% concordance with clinical outcomes (p < 0.001).
From a market perspective, the thyroid diagnostics space is undergoing a quiet revolution. Global prevalence of thyroid disorders is rising (driven by iodine deficiency, autoimmune diseases, and aging populations), pushing demand for affordable, scalable tests. Meanwhile, precision medicine initiatives (e.g., tailoring levothyroxine doses to TSH thresholds) require assays with tight reference intervals—something the KTE6905 delivers, with pre-defined ranges for adults, children, and pregnant women (trimester-specific cutoffs included). Abbkine’s edge? Per-test pricing at 4.50 (vs. 7–10 for CLIAs) and a “no-questions-asked” replacement policy for expired/broken kits—critical for budget-strapped clinics in low-resource settings.
Looking ahead, the role of TSH in precision medicine is expanding. Emerging research links TSH variability to cardiovascular risk (even within “normal” ranges), neurodegenerative diseases (Alzheimer’s), and fertility outcomes—creating demand for assays that track TSH dynamics over time. The EliKine™ KTE6905 is positioned to lead here: its compatibility with automated liquid handlers (via pre-coated strips) enables large-scale cohort studies, while Abbkine’s roadmap includes a “TSH-Stimulated T4” companion kit to assess thyroid reserve—addressing a key gap in current diagnostics. For labs aiming to future-proof their thyroid testing, this kit isn’t just a tool; it’s a strategic investment.
In summary, the EliKine™ Human TSH ELISA Kit (KTE6905) by Abbkine isn’t another “me-too” assay—it’s a response to the unmet needs of clinicians and researchers tired of compromise. With best-in-class sensitivity, ironclad specificity, and a workflow designed for real-world efficiency, it transforms TSH measurement from a bottleneck into a competitive advantage. Whether you’re running a busy diagnostic lab, unraveling thyroid-autonomic crosstalk in a research project, or developing the next generation of thyroid therapies, this kit delivers the reliability you need to move forward with confidence.
Ready to elevate your TSH testing? Explore the EliKine™ Human TSH ELISA Kit (KTE6905) and its full validation data—including sample type compatibility, reference intervals, and case studies—at the https://www.abbkine.com/product/elikine-human-tsh-elisa-kit-kte6905/.











