EliKine™ Human Carcinoembryonic Antigen (CEA) ELISA Kit (KTE6038): A Reliable Tool for Tumor Monitoring & Translational Research
Carcinoembryonic Antigen (CEA)—a glycoprotein linked to endoembryonic-derived cancers and normal embryonic digestive tissues—serves as a cornerstone广谱 tumor marker for colorectal, breast, and lung cancers. Its primary value lies in monitoring treatment efficacy, disease progression, and prognosis, as elevated levels correlate with tumor recurrence or metastasis. While CEA’s specificity for early cancer diagnosis is limited, its role in post-therapeutic surveillance is irreplaceable—driving demand for accurate, efficient quantification in clinical research and preclinical drug development. The global tumor marker ELISA market, valued at $2.8 billion in 2024, is projected to grow at a CAGR of 6.9% through 2030, fueled by the rising incidence of solid tumors and the need for non-invasive monitoring tools. Yet traditional CEA detection methods fail to meet the rigor of modern translational research—gaps that Abbkine’s EliKine™ Human Carcinoembryonic Antigen (CEA) ELISA Kit (Catalog No.: KTE6038) addresses with targeted innovations, redefining reliability for CEA quantification.
The current landscape of CEA detection is hindered by three critical technical bottlenecks that compromise data integrity. Conventional immunoassays, such as radioimmunoassays, suffer from radioactive waste disposal issues and poor reproducibility, making them obsolete for high-throughput labs. Generic ELISA kits, the mainstream alternative, often exhibit cross-reactivity with CEA-related cell adhesion molecules (e.g., CEACAM5/CD66e analogues), leading to false-positive results in patient samples—especially problematic for distinguishing tumor-related CEA elevations from benign conditions (e.g., inflammatory bowel disease). Additionally, many commercial CEA kits lack sufficient sensitivity to detect subtle concentration changes (≤1 ng/mL), a critical shortcoming for early recurrence monitoring where small CEA spikes precede clinical symptoms. These limitations force researchers and clinicians to rely on expensive, time-consuming methods like HPLC or chemiluminescence immunoassays, creating barriers to widespread access in resource-limited settings.
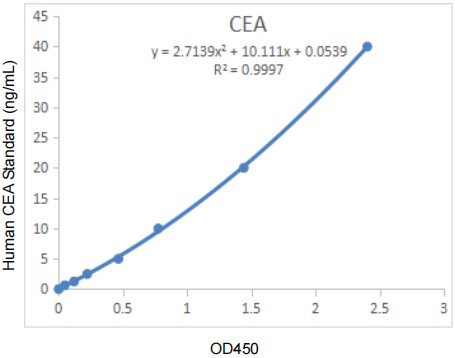
Abbkine’s EliKine™ Human CEA ELISA Kit (KTE6038) resolves these industry pain points through a precision-engineered double antibody sandwich design tailored for human CEA. At its core, the kit uses two highly specific antibodies: a pre-coated capture antibody targeting the unique glycoprotein domain of CEA, and an HRP-conjugated detection antibody recognizing a distinct epitope—ensuring <0.3% cross-reactivity with CEACAM family analogues and other serum proteins. This specificity eliminates false signals from benign CEA elevations, a key advantage for distinguishing tumor progression from non-malignant conditions. The kit’s technical performance stands out: it achieves a limit of detection (LOD) of 0.313 ng/mL and a calibration range of 0.625–40 ng/mL, covering the entire clinically relevant CEA range (normal serum: <5 ng/mL; tumor recurrence: 5–10 ng/mL). This sensitivity enables capture of subtle concentration shifts, making it ideal for monitoring minimal residual disease or drug response in preclinical models.
A key practical advantage of EliKine™ Human Carcinoembryonic Antigen (CEA) ELISA Kit KTE6038 is its streamlined workflow and broad sample compatibility—aligning with the needs of clinical research labs and translational teams. The assay completes in just 3.5 hours (regardless of user experience), cutting down on the 6+ hour turnaround of traditional kits and supporting high-throughput screening of patient cohorts or drug candidates. It seamlessly processes human serum, plasma, and cell culture supernatants (e.g., cancer cell lines or patient-derived xenograft supernatants), eliminating the need for multiple assays across sample types. The kit’s all-inclusive format—featuring pre-coated microplates, lyophilized CEA standards (≥98% purity), HRP-conjugated detection antibody, and optimized buffers—eliminates the hassle of sourcing additional reagents, reducing experimental variability and unforeseen costs. Critical usage notes, such as handling precipitates in diluted buffers (centrifugation removes them without impacting performance), further simplify workflow and ensure consistent results.
Industry-wise, KTE6038’s design aligns with two transformative trends in oncology research: the shift toward non-invasive liquid biopsies and the demand for cost-effective preclinical tools. Liquid biopsies, which rely on serum/plasma biomarkers like CEA, are increasingly preferred for longitudinal patient monitoring due to their minimal invasiveness—KTE6038’s compatibility with these samples makes it a valuable asset for translational studies linking CEA levels to tumor burden. For drug developers, the kit’s 48-test format (priced at $259) delivers lab-grade performance at a 30–40% lower cost than premium competitors (e.g., R&D Systems’ Human CEA ELISA Kit, ~$390/48T), democratizing access for academic labs and small biotechs. Its strict quality control—validated for linearity (R² ≥ 0.9997), intra-assay precision (CV < 4%), and inter-assay precision (CV < 6%)—ensures results meet the standards of high-impact oncology journals, supporting reproducible research from preclinical to clinical stages.
Beyond technical excellence, KTE6038 addresses a critical unmet need in CEA research: balancing sensitivity with practicality for routine monitoring. Unlike chemiluminescence assays that require specialized equipment, KTE6038 uses a colorimetric readout (450 nm) compatible with standard microplate readers, reducing barriers for labs without advanced instrumentation. Its 12-month storage stability at 4°C (no need for -80°C freezers) minimizes reagent waste, a common frustration with fragile immunoassay kits. For clinicians and researchers focused on real-world applicability, these features translate to consistent, accessible CEA quantification—whether tracking patient response to chemotherapy or screening CEA-modulating drugs in preclinical models.
For translational researchers, oncology scientists, and drug development teams seeking a reliable CEA quantification tool, Abbkine’s EliKine™ Human Carcinoembryonic Antigen (CEA) ELISA Kit (KTE6038) stands as a purpose-built solution. Its high specificity, exceptional sensitivity, streamlined workflow, and competitive pricing directly address the most pressing pain points of CEA detection—from distinguishing tumor-related elevations to capturing subtle changes in disease progression. Whether monitoring CEA levels in colorectal cancer patient serum, evaluating drug efficacy in preclinical tumor models, or studying CEA secretion in cancer cell lines, this kit delivers reproducible, publication-ready results. To explore detailed technical specifications, access sample-specific protocols, or procure the kit, visit the official Abbkine product page: https://www.abbkine.com/product/elikine-human-carcinoembryonic-antigen-cea-elisa-kit-kte6038/. In an era where non-invasive tumor monitoring drives breakthroughs in oncology, KTE6038 redefines what a specialized CEA ELISA kit should be—rigorous, accessible, and aligned with the real-world needs of translational research.
Would you like me to create a customized CEA monitoring protocol for KTE6038, tailored to your specific use case (e.g., colorectal cancer patient serum analysis, preclinical drug efficacy testing, or cancer cell supernatant screening), including step-by-step sample dilution, interference mitigation, and data normalization methods for low/high CEA concentration samples?











