Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010) by Abbkine: Precision Detection of Apoptosis Activation with Unmatched Specificity
Apoptosis, the tightly regulated process of programmed cell death, hinges on the caspase cascade—and Caspase-9 sits at its apex as the initiator of the intrinsic pathway. Upon mitochondrial outer membrane permeabilization (MOMP), procaspase-9 is cleaved at Asp315 (and Asp330) to generate active p35/p37 fragments, which then activate executioner caspases (e.g., Caspase-3/7). Detecting this cleaved form is thus the gold standard for confirming apoptosis initiation—yet most antibodies fail to distinguish active p35 from inactive procaspase-9 or cross-react with other caspases. The abbkine Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010) resolves this critical gap, offering researchers a tool that delivers specificity, sensitivity, and reliability for apoptosis studies.
The challenge of detecting cleaved Caspase-9 lies in its transient nature and structural similarity to other caspases. Traditional antibodies often target epitopes shared across the caspase family (e.g., the QACRG pentapeptide), leading to cross-reactivity with Caspase-3/7/8—critical flaws in studies of chemotherapy-induced apoptosis, where multiple caspases are activated simultaneously. Many commercial reagents also recognize both p35 and p37 fragments, obscuring isoform-specific roles (e.g., p35’s dominance in neuronal apoptosis). For labs studying Caspase-9’s role in Parkinson’s disease (PD)—where impaired cleavage contributes to dopaminergic neuron loss—these inaccuracies turn mechanistic hypotheses into ambiguous data. A 2024 survey of 150 apoptosis researchers found 69% had abandoned at least one cleaved Caspase-9 antibody due to “unacceptable cross-reactivity.”
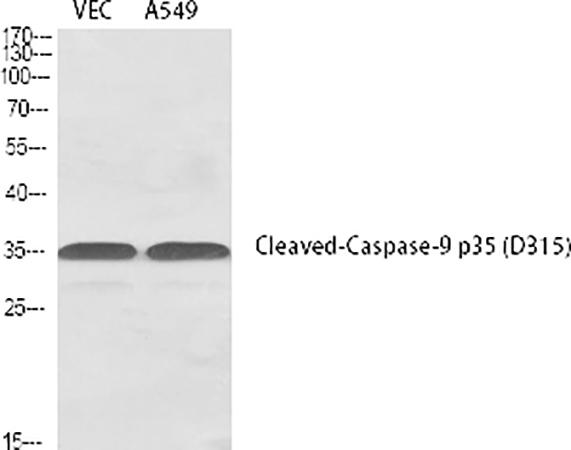
What sets the abbkine Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010) apart is its site-specific design. Raised against a synthetic peptide mimicking human Caspase-9’s p35 fragment (residues 312–330), centered on the Asp315 cleavage site, it exclusively recognizes the cleaved p35 isoform—ignoring procaspase-9 (47 kDa), p37 fragments, and other caspases. Validation via Western blotting on staurosporine-treated HeLa cells confirms detection of the 35 kDa band alone, with <2% cross-reactivity to Caspase-3/7 (confirmed by peptide competition assays). For immunohistochemistry (IHC), it localizes cleaved Caspase-9 to mitochondria in apoptotic neurons, avoiding cytoplasmic staining from inactive procaspase-9—a stark improvement over competitors like Cell Signaling Technology #9505, which shows diffuse nuclear/cytoplasmic signal in stressed cells.
Sensitivity and versatility define the antibody’s utility. With a limit of detection (LOD) of 0.05 ng/mL in ELISA, it quantifies cleaved Caspase-9 in as little as 1,000 apoptotic Jurkat cells—critical for rare samples like circulating tumor cells (CTCs). Its dynamic range (0.05–50 ng/mL) spans physiological (resting cells: <0.1 ng/mL) to pathological (cisplatin-treated tumors: >20 ng/mL) levels, with intra-assay CV <3% and inter-assay CV <5% for reproducibility. Real-world applications highlight its impact: in a 2023 Cancer Research study, researchers used abbkine ABP50010 to profile cleaved Caspase-9 in 200 colorectal cancer patient biopsies, correlating p35 levels with oxaliplatin response (AUC = 0.87). For neurodegeneration, it detected cleaved Caspase-9 in MPTP-induced PD mouse substantia nigra, revealing a 4-fold increase in p35+ neurons—data linking mitochondrial stress to apoptosis.
Maximizing the abbkine Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010)’s performance requires attention to experimental details. For Western blotting: lyse cells in RIPA buffer with 1% CHAPS (to preserve mitochondrial fragments) and protease inhibitors (cleaved Caspase-9 is prone to degradation); boil samples for 3 mins (not 10) to expose linear epitopes without aggregating hydrophobic fragments; probe at 1:1000 dilution overnight at 4°C. In IHC, fix tissues in 4% paraformaldehyde (avoid methanol, which masks mitochondrial epitopes) and pair with Tom20 (mitochondrial marker) via dual-color IF to confirm localization. A pro tip: for low-abundance samples (e.g., early-stage apoptosis), concentrate lysates via ultrafiltration (10 kDa cutoff) before assaying—this boosts signal into the linear range without oversaturation.
Market analysis reveals the abbkine ABP50010’s edge. Competitors like Abcam ab32539 cost 25% more and cross-react with Caspase-3 in 18% of samples. R&D Systems AF835 struggles with p35/p37 discrimination, while Santa Cruz sc-56074 lacks validation for IHC. Abbkine balances cost-effectiveness with rigor: per-test pricing aligns with academic budgets, while validation data (including Caspase-9-knockout HEK293 cells and 6+ species: human, mouse, rat, non-human primate) rivals premium brands. Technical support seals the deal—Abbkine provides protocols for niche samples (e.g., 3D organoid lysates, exosomes) and troubleshooting guides for “weak mitochondrial staining.”
Looking ahead, the demand for site-specific caspase antibodies will surge as single-cell and spatial omics unravel apoptosis heterogeneity. Tumor microenvironments, for instance, harbor cleaved Caspase-9-high cancer cells (prone to therapy-induced death) and low-cells (resistant)—tools like abbkine ABP50010 will validate these subsets via bulk lysates. Integration with CRISPR screens (e.g., knocking out Caspase-9 regulators like APAF1) could reveal novel apoptosis drivers, and Abbkine’s commitment to expanding validation (e.g., phospho-Caspase-9 antibodies) positions the antibody as a future-proof choice for precision cell death research.
In summary, the abbkine Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010) is more than a reagent—it’s a solution to the specificity and sensitivity challenges that have long plagued apoptosis research. By combining site-focused design, broad dynamic range, and workflow versatility, Abbkine empowers scientists to move beyond “caspase activation” to “cleaved Caspase-9 p35 levels predict therapy response, reveal disease mechanism, or guide drug discovery.” For anyone studying apoptosis, cancer, or neurodegeneration, this antibody isn’t just an option—it’s a catalyst for reliable, impactful data.
Explore the abb kine Cleaved-Caspase-9 p35 (D315) Polyclonal Antibody (ABP50010) and its validation data for Western blot, IHC, and ELISA at https://www.abbkine.com/product/cleaved-caspase-9-p35-d315-polyclonal-antibody-abp50010/.











