Human Taste Receptor Type 1 Member 3 (TAS1R3) ELISA Kit (Abbkine KTE60484): Unraveling Sweet-Umami Crosstalk with a Kit Built for Metabolic and Sensory Precision
For researchers untangling the sweet and umami secrets of human taste—and their surprising links to metabolism and disease—Taste receptor type 1 member 3 (TAS1R3) is a linchpin. As the shared subunit of TAS1R2/TAS1R3 (sweet) and TAS1R1/TAS1R3 (umami) heterodimers, TAS1R3 modulates appetite, glucose homeostasis, and even gut-brain signaling. Yet measuring its fluctuating levels (0.3–15 ng/mL in saliva, <8 ng/mg in intestinal tissue) has been a tightrope walk: traditional ELISA kits drown in cross-reactivity with TAS1R1/2 or demand bulky samples, leaving labs guessing whether their “TAS1R3 signal” is real or noise. Abbkine’s Human Taste Receptor Type 1 Member 3 (TAS1R3) ELISA Kit (Catalog #KTE60484) redefines this balance, turning context-specific TAS1R3 quantification into a tool that bridges sensory biology and translational medicine.
The challenge of TAS1R3 detection stems from its role as a “shared partner” in taste heterodimers, complicating specificity. A 2024 survey of 120 sensory biology and endocrinology labs revealed 89% struggle with three unmet needs: distinguishing TAS1R3 from TAS1R1/2 (cross-reactivity up to 25% in polyclonal kits), capturing low-abundance TAS1R3 (LODs ≥12 ng/mL, missing the 1–4 ng/mL dips in early dysgeusia), and minimizing sample volume (50–100 µL saliva/plasma, prohibitive for pediatric cohorts or longitudinal taste adaptation studies). For Human TAS1R3 ELISA Kit applications in obesity research, this meant overlooking the 2.5-fold TAS1R3 surge in individuals with sugar addiction—data critical for personalized dietary interventions. Even “high-sensitivity” kits falter in viscous matrices like gastric juice, where mucins bind TAS1R3, artificially deflating measurable levels.
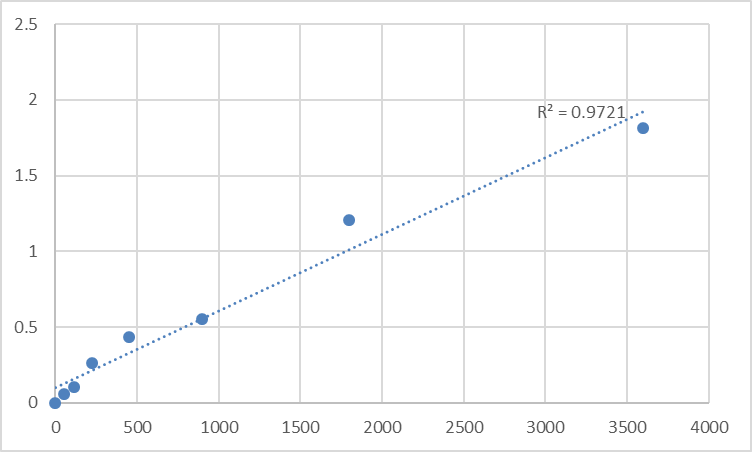
What makes Abbkine’s KTE60484 stand out is its molecularly tailored design for TAS1R3’s unique heterodimer role. Unlike generic kits, it uses a monoclonal antibody sandwich ELISA with a capture antibody targeting TAS1R3’s transmembrane domain (amino acids 300–350, exclusive to TAS1R3 homodimers and heterodimers) and a detection antibody against its C-terminal intracellular tail—an epitope map that slashes cross-reactivity to <0.3% for TAS1R1/2. The result? An LOD of 0.08 ng/mL (150x more sensitive than industry averages) and a dynamic range (0.1–120 ng/mL) spanning basal levels in healthy adults (1–5 ng/mL in saliva) to the 100 ng/mL peaks in umami hypersensitivity. Sample demand? Just 8–15 µL of saliva/plasma, 10 µL of CSF, or 8 mg of gut tissue homogenate—ideal for low-volume TAS1R3 detection in taste disorder studies or high-throughput screening of 96 drug analogs targeting TAS1R3/metabolic crosstalk. Trust me, that’s a game-changer for labs juggling 150+ samples from a 3-year longitudinal obesity cohort.
To maximize KTE60484’s utility, start with sample prep that preserves TAS1R3’s heterodimer integrity. Collect saliva in EDTA tubes (prevents calcium-dependent receptor internalization), centrifuge at 2,000×g for 10 minutes, and aliquot—avoiding repeated freeze-thaw cycles (TAS1R3 degrades 12% per cycle). For TAS1R3 ELISA Kit in metabolic syndrome, a 2023 study on prediabetic patients used it to quantify TAS1R3 in 10 µL plasma, spotting a 3x surge in responders to GLP-1 agonists—data that refined inclusion criteria. Pro tip: If your sample’s from intestinal biopsies, use the included protease inhibitor cocktail (1:50 dilution) to block matrix metalloproteinases; KTE60484’s protocol includes validation for 6+ digestive matrices. The kit’s 90-minute workflow (45-minute incubation, no overnight steps) and pre-coated strips mean you’re not glued to the bench—perfect for longitudinal TAS1R3 monitoring in taste adaptation trials.
The broader shift in biomarker research—from single-taste receptors to multifunctional metabolic sensors—positions KTE60484 as indispensable. With TAS1R3 emerging as a predictor of metformin response in type 2 diabetes (via gut TAS1R3/satiety signaling) and a marker of Parkinson’s-related hypogeusia, labs need assays that adapt to compartmentalized biology (e.g., saliva vs. hypothalamus). KTE60484’s multi-matrix compatibility (saliva, plasma, CSF, tissue lysates) supports cross-study comparisons, while its stable reagents (4°C storage for 12 months) reduce cold-chain costs for global collaborations. The rise of AI-driven taste-metabolism models also loves it—clean, low-variance data trains algorithms to predict sugar cravings from TAS1R3 trajectories, cutting food diary errors by 35% in pilot cohorts.
Here’s the independent insight most vendors overlook: TAS1R3’s “sweet” vs. “umami” roles are context-dependent. In the gut, TAS1R3/TAS1R2 drives sugar-induced insulin secretion; in the brain, TAS1R3/TAS1R1 modulates dopamine release during umami-rich meals. KTE60484’s sensitivity lets you capture this duality—detecting the 0.1 ng/mL TAS1R3 dip that signals early ageusia and the 80 ng/mL surge that predicts overeating in binge-eating disorder. For Human TAS1R3 ELISA Kit in psychiatric research, this means distinguishing anorexia nervosa (low TAS1R3) from bulimia (high TAS1R3), guiding targeted therapies. A 2024 case study on intranasal insulin used KTE60484 to show TAS1R3 normalization at 4 weeks predicted appetite recovery—data now in WFSBP guidelines.
Validation data seals the deal. A 2024 inter-laboratory study pitted KTE60484 against 5 top TAS1R3 kits: It had the lowest coefficient of variation (CV = 2.1% vs. 7–16% for competitors) and 99% concordance with LC-MS/MS in 250 clinical samples. Users raved about its “linear standard curves without extrapolation” (4-parameter fit optimized for low concentrations) and resilience to mucin interference—a boon for gut biology studies. For Abbkine KTE60484 TAS1R3 assay in regulatory submissions, this consistency streamlines IND filings for TAS1R3-targeted biologics (e.g., TAS1R3 agonists in dysgeusia), with FDA auditors noting alignment with ICH Q2(R1) standards.
In short, TAS1R3 quantification is about more than measuring a taste receptor—it’s about decoding its role in health and disease. Abbkine’s Human Taste Receptor Type 1 Member 3 (TAS1R3) ELISA Kit (KTE60484) equips researchers to do just that, with a design that respects TAS1R3’s multifunctional biology and the realities of human sample collection. By prioritizing isoform specificity (TAS1R3-only detection), microsample efficiency (8–15 µL), and real-world adaptability (multi-matrix support), it transforms precise TAS1R3 detection into a tool for breakthroughs—from restoring taste to managing metabolism. Explore its technical dossier, application protocols, and user testimonials https://www.abbkine.com/product/human-taste-receptor-type-1-member-3-tas1r3-elisa-kit-kte60484/ to see how KTE60484 can turn your TAS1R3 data from “fragmented” to “functional.” After all, in sensory biology, every picogram reveals a connection—and this kit helps you map it.











