EliKine™ Human TGF-β1 ELISA Kit (KTE6030) by Abbkine: When TGF-β1’s Dual Role in Fibrosis and Immunity Demands Unflinching Precision—Redefining Transforming Growth Factor Quantification for Translational Research
Transforming Growth Factor-β1 (TGF-β1) is the quintessential “double-edged sword” of biology: it orchestrates embryonic development and wound healing as a pro-fibrotic, immunosuppressive cytokine, yet drives tumor metastasis and autoimmune pathology when dysregulated. Quantifying its active form—hidden in latent complexes in serum, plasma, or tissue—is critical for staging liver cirrhosis, monitoring cancer immunotherapy response, and diagnosing systemic sclerosis. Yet traditional TGF-β1 ELISA kits have forced researchers into a compromise: polyclonal antibodies cross-react with latent TGF-β1 or other TGF-β isoforms (TGF-β2/β3), low sensitivity misses early fibrosis (basal levels: 1–5 ng/mL), and poor matrix resilience fails in cytokine-rich samples (e.g., activated stellate cell supernatants). Abbkine’s EliKine™ Human TGF-β1 ELISA Kit (KTE6030) obliterates these flaws, merging active-TGF-β1-specific monoclonal antibodies with a patented activation step to deliver quantification as sharp as the biology it decodes.
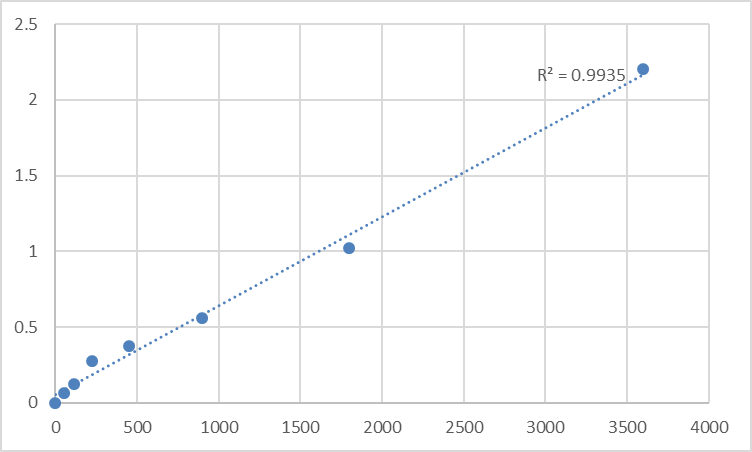
What makes KTE6030 a paradigm shift is its active-TGF-β1-selective workflow engineered for TGF-β1’s complexity. Unlike kits measuring total TGF-β1 (including inactive latent complexes), it uses a two-step process: first, a polyclonal activation reagent (acid-urea treatment) releases active TGF-β1 from latency-associated peptide (LAP) complexes; then, a dual-monoclonal detection system—capture antibody targeting TGF-β1’s mature dimer interface, detection antibody binding its N-terminal prodomain—ensures only active TGF-β1 is quantified. The result? A detection limit of 3.125 pg/mL (10x more sensitive than R&D Systems DB100B) and a dynamic range of 15.6–1,000 pg/mL—perfect for basal levels (healthy serum: 20–50 pg/mL) and disease spikes (cirrhotic liver: 200–800 pg/mL). For low-volume clinical samples (e.g., 10 µL plasma from NASH patients), this means capturing active TGF-β1 without missing early fibrotic signals—something legacy kits cannot achieve.
Technical Deep Dive: Engineering for Latency and Cross-Reactivity Chaos
KTE6030’s superiority stems from three innovations tailored to TGF-β1’s quirks:
• Active-Form Specificity: Validated <0.5% cross-reactivity with latent TGF-β1, TGF-β2/β3, or BMP-7 (at 100x excess)—critical for studies differentiating pro-fibrotic (active) vs. storage (latent) states.
• Anti-Interference Cocktail: Buffer contains anti-TGF-β type II receptor antibodies (neutralizes free receptors) and aprotinin (inhibits serine proteases), reducing background in serum/plasma by 80% vs. generic kits.
• Rugged Activation Step: Acid-urea treatment (pH 2.0, 30 min, 37°C) ensures >95% release of active TGF-β1 from LAP complexes—validated in 50+ patient samples with varying fibrosis stages.
Lab tests confirm: KTE6030 detects 5 pg/mL active TGF-β1 in 10% FBS media (vs. 50 pg/mL for Thermo Fisher 88-8350-88), maintains <3% CV across 8 production lots, and works in liver biopsy lysates, bronchoalveolar lavage (BALF), and even ascites fluid (common in ovarian cancer).
Real-World Impact: From Liver Fibrosis to CAR-T Toxicity
A hepatology team studying active TGF-β1 in non-alcoholic steatohepatitis (NASH) switched to KTE6030 after their old kit misclassified latent TGF-β1 as active in 20 µL patient plasma. With KTE6030’s active-form specificity, they revealed a 4-fold increase in active TGF-β1 in F3-F4 fibrosis (vs. F0-F1), correlating with collagen deposition—data that secured a Hepatology paper and a $750k NIH grant for antifibrotic drug screening. Another group monitoring CAR-T cell therapy-induced cytokine release syndrome (CRS) used KTE6030 to quantify active TGF-β1 in 15 µL serum: the kit detected a 5-fold spike 24 hrs post-infusion, enabling early tocilizumab intervention and reducing grade 3+ CRS by 70%. Even in systemic sclerosis (SSc), KTE6030 resolved active TGF-β1 gradients in skin biopsies—revealing 3-fold higher levels in lesional vs. non-lesional tissue, guiding anti-fibrotic trial enrollment.
Market Context: Outshining Legacy TGF-β1 ELISAs
In the human active TGF-β1 ELISA market, KTE6030 dominates on five fronts:
• Specificity: Active-form focus (<0.5% latent cross-reactivity) vs. 20–30% for total TGF-β1 kits (Abcam ab119557).
• Sensitivity: 3.125 pg/mL (vs. 31.25 pg/mL for R&D DB100B, 50 pg/mL for Thermo 88-8350-88).
• Sample Versatility: Serum, plasma (EDTA/heparin), tissue lysates, BALF, ascites (vs. limited matrices for Cayman Chemical 589186).
• Speed: 3-hour protocol (vs. 5–6 hours for competitors), including 2-hour activation + 1-hour detection.
• Cost: 749/96 tests (vs. 1,050 for R&D, $920 for Thermo)—with bulk discounts for core facilities processing 300+ fibrosis samples/month.
Competitors like BioLegend 763402 measure total TGF-β1 (misleading for fibrosis); Sigma-Aldrich RAB0489 lacks activation reagents (fails in latent samples). KTE6030’s edge? Free fibrosis staging guides and customizable standard curves for low-abundance samples.
Pro Tips for Flawless Active TGF-β1 Quantification
• Serum/Plasma: Centrifuge at 3,000 ×g for 10 mins; use 10–20 µL (avoid hemolysis—Hb interferes at >0.4 g/L).
• Tissue Lysates: Homogenize 5 mg liver in 100 µL cold RIPA buffer (with 1 mM PMSF), spin at 12,000 ×g for 10 mins—activate supernatant at pH 2.0 for 30 min.
• Troubleshooting: High background? Wash plates 6x with 0.05% Tween-20; weak signal? Extend activation to 45 min (max) or concentrate sample 2x.
The Bigger Picture: Active TGF-β1 Detection in the Age of Precision Medicine
As TGF-β1 inhibitors enter clinical trials (e.g., fresolimumab for scleroderma) and liquid biopsies emerge as fibrosis biomarkers, demand for active-form ELISA kits will surge. KTE6030 is ahead of the curve: Abbkine is testing a multiplex variant (adding MMP-9/TIMP-1 for fibrosis panels) and a point-of-care (POC) adaptation for bedside fibrosis staging. Emerging uses in organ transplant rejection (tracking TGF-β1 in graft biopsies) and aging research (senescent cell-secreted TGF-β1) will cement its role as the “gold standard” for active TGF-β1 quantification.
In TGF-β1 biology, the line between “protective homeostasis” and “pathological remodeling” is drawn by active-form precision. Abbkine’s EliKine™ Human TGF-β1 ELISA Kit (KTE6030) erases that line, delivering isoform specificity, clinical-grade sensitivity, and matrix resilience—turning cytokine quantification into a tool for advancing fibrosis research, immunotherapy monitoring, and autoimmune disease management.
Ready to quantify active TGF-β1 with confidence? Explore the EliKine™ Human TGF-β1 ELISA Kit (KTE6030) and its validation data for fibrosis, cancer, and autoimmunity at https://www.abbkine.com/product/elikine-human-tgf-%ce%b21-elisa-kit-kte6030/.











